Product Offerings
Customizable OptiDropperTM
Ophthalmic Dropper Bottles
Select a Bottle

Part Number
EA Per Box
Total EA
Unit Price
Price
Select a Tip or Tip/Cap
Part Number
EA Per Box
Total EA
Unit Price
Price
Select a Cap

Part Number
EA Per Box
Total EA
Unit Price
Price
Frequently Asked Questions
What final release testing is performed for these bottle products?
| Test | Method | Component |
| Sterility | Direct Transfer Method Embedded Spore Strip accredited to the ISO 17025 | Bottle, Tip, Cap |
| Endotoxin/LAL | ISO 17025 – (USP), General Chapter <85>, (USP), General Chapter <161>, Medical Devices | Bottle, Tip, Cap |
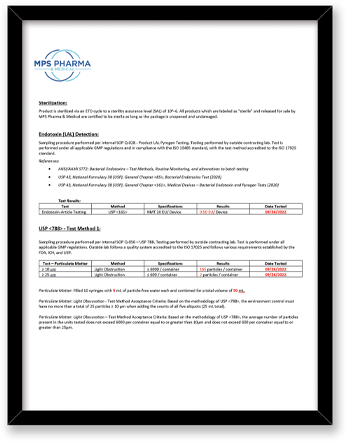
| USP <788> Test Method 1 – Particulates | Test Method Acceptance Criteria: Based on the methodology of USP <788> | Bottle, Tip (Testing limited to fluid contact surfaces) |
All testing is performed by qualified third party testing labs contracted with MPS Pharma. The final measurements are all reported on our current Certificate of Analysis.
Why do you measure particulates to USP <788>?
USP <771> Ophthalmic Products – Quality Tests:
“Two general categories apply for product administration to the tissues in the eye. Intraocular administration includes all ophthalmic products that cross (penetrate) boundary tissue, such as the cornea and sclera. For subvisible content, USP guidance is followed. Products for intraocular use must comply with Particulate Matter in Ophthalmic Solutions <789>. Product for extraocular use must comply with Particulate Matter in Injections <788>.”
Both USP <788> and <789> are applicable to finished drug products; containers are not considered finished drug product. The testing MPS Pharma has performed on the bottles is based on the methodology of USP <788>, with particulate measurements reported for 10 um and 25 um sizes against limits reported on a per container basis.
Can I see a sample Certificate of Analysis that comes with this product?
See the attached PDF for a sample Certificate of Analysis
I have a special requirement (new pack size, special drop size, etc). Can you help?
Are the parts interchangeable? How does the numbering system work on these parts?
All items with a UA8-425 pre-fix are compatible with each other.
All items with a UA15-415 pre-fix are compatible with each other.
UA8-425 & UA15-415 items are not compatible with each other.
If I open the pouch, will the bottle contents still be sterile?
What is the drop size for these bottles?
A general water reference point can be provided to our customers as a starting guideline:
| Size(s) | Drop Size |
| 3 mL | 42 ul +/- 5 uL |
| 7, 10, 15, 30 mL | 40 ul +/- 5 uL |
What are the bottles made of? Can more information be shared?
A more detailed material profile is available to share with customers who complete a non-disclosure agreement.
Are dimensional drawings of the bottle available?
Do you carry bottles that offer UV protection? Is there data available to share?
Specific testing standards surrounding UV protection of solutions are always performed using the container closure system itself plus the drug contents. MPS does not provide drug with the bottle, therefore no meaningful data can be provided that our end user pharmacies may leverage for their own purposes.
We can anecdotally report however that our customers do routinely use specific products of ours for light protected solutions. Those same customers do report the bottle, in combination with their drug product, pass the UV protection requirement as defined in the existing testing standards.
Please reach out to us to find out which items would work best in supporting a successful passage of these tests.
Can I get a specification sheet of this item for my records?
See the attached document for the specification sheet
Our pharmacy follows USP <797> and it requires we use depyrogenated materials. Are your products depyrogenated? Do you have a Certificate of Analysis (C of A) stating such?
All MPS lots are tested for endotoxins (pyrogens) and will meet the materials requirements as defined in USP <797>. A detailed explanation of this can be found in this PDF link. All MPS C of As include measurements for these endotoxins.
Have any questions?
Contact our Customer Service team for additional support at
(844) 641-3814 or by email at sales@mpspharma-inc.com

 0
0